Abstract
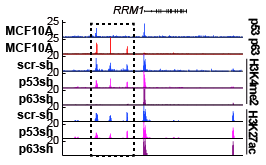
Transcriptional activation by p53 provides powerful, organism-wide tumor suppression. We hypothesized that the local chromatin environment, including differential enhancer activities, contributes to various p53-dependent transcriptional activities in different cell types during stress-induced signaling. In this work, using ChIP-seq, immunoblotting, qPCR, and computational analyses across various mammalian cell lines, we demonstrate that the p53-induced transcriptome varies by cell type, reflects cell type-specific activities, and is considerably broader than previously anticipated. We found that these molecular events are strongly influenced by p53’s engagement with differentially active cell typespecific enhancers and promoters. We also observed that p53 activity is dependent on the p53 family member tumor protein p63 in epithelial cell types. Notably, we demonstrate that p63 is required for epithelial enhancer identity including enhancers used by p53 during stress-dependent signaling. Loss of p63, but not p53, caused site-specific depletion of enhancer-associated chromatin modifications, suggesting that p63 functions as an enhancer maintenance factor in epithelial cells. Additionally, a subset of epithelial-specific enhancers is dependent on the activity of p63 providing a direct link between lineage determination and enhancer structure. These results suggest that a broad, cellintrinsic mechanism controls p53-dependent cellular stress response through differential regulation of cis-regulatory elements.