Abstract
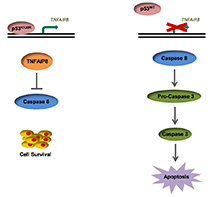
The p53 tumor suppressor gene encodes a sequence-specific transcription factor. Mutations in the coding sequence of p53 occur frequently in human cancer and often result in single amino acid substitutions (missense mutations) in the DNA binding domain (DBD), blocking normal tumor suppressive functions. In addition to the loss of canonical functions, some missense mutations in p53 confer gain-of-function (GOF) activities to tumor cells. While many missense mutations in p53 cluster at six “hotspot” amino acids, the majority of mutations in human cancer occur elsewhere in the DBD and at a much lower frequency. We report here that mutations at K120, a non-hotspot DNA contact residue, confer p53 with the previously unrecognized ability to bind and activate the transcription of the pro-survival TNFAIP8 gene. Mutant K120 p53 binds the TNFAIP8 locus at a cryptic p53 response element that is not occupied by wild-type p53. Furthermore, induction of TNFAIP8 is critical for the evasion of apoptosis by tumor cells expressing the K120R variant of p53. These findings identify induction of pro-survival targets as a mechanism of gain-of-function activity for mutant p53 and will likely broaden our understanding of this phenomenon beyond the limited number of GOF activities currently reported for hotspot mutants.