Abstract
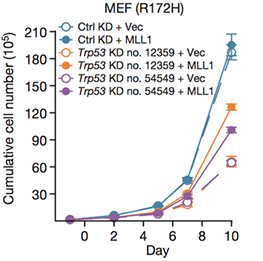
TP53 (which encodes p53 protein) is the most frequently mutated gene among all human cancers. Prevalent p53 missense mutations abrogate its tumour suppressive function and lead to a ‘gain-of-function’ (GOF) that promotes cancer. Here we show that p53 GOF mutants bind to and upregulate chromatin regulatory genes, including the methyltransferases MLL1 (also known as KMT2A), MLL2 (also known as KMT2D), and acetyltransferase MOZ (also known as KAT6A or MYST3), resulting in genome-wide increases of histone methylation and acetylation. Analysis of The Cancer Genome Atlas shows specific upregulation of MLL1, MLL2, and MOZ in p53 GOF patient-derived tumours, but not in wild-type p53 or p53 null tumours. Cancer cell proliferation is markedly lowered by genetic knockdown of MLL1 or by pharmacological inhibition of the MLL1 methyltransferase complex. Our study reveals a novel chromatin mechanism underlying the progression of tumours with GOF p53, and suggests new possibilities for designing combinatorial chromatin-based therapies for treating individual cancers driven by prevalent GOF p53 mutations.